Note: this essay could not have been written without the help of Aishwarya Khanduja and a few other anonymous people who have worked in the pathology startup scene. Also, another reminder about the April 16th NYC bio-ML meetup!
Introduction
Back when I was in college (2015-2019), I wasn’t super tuned into how AI was altering biology research. In many ways, it still wasn’t. Alphafold2 wasn’t released, the earliest ‘let’s create drugs with AI’ companies were still either starting or failing, and the scale hypothesis had yet to fully be realized. A dark era!
But what I was tuned in to was how AI was altering the ‘pre-existing medical data’ research field. It felt like the obvious place ML could be easily applied. After all, terabytes of data here were collected month-after-month, just as an incidental result of patients going through the medical system. Data here came ‘free’; no need to spend millions of dollars to acquire new datasets, no need to hire annotators. In the absolute earliest form, this was X-rays, MRI scans, ultrasound, electronic health records, and the like.
But, for a long time, pathology was left out of this. This was because it initially lacked the same level of digitization as radiology or electronic health records. Unlike those modalities, which had been stored in digital formats for decades, pathology slides were still predominantly physical, analyzed underneath a microscope and then discarded. This was for a decent reason: pathology slides are huge if you want to account for the massive resolution that microscopes are capable of. On average, each slide could be on the order of gigabytes of size, and hospitals, lab centers, and universities likely would collect hundreds, if not thousands, of these slides per day. Moreover, they required expensive whole-slide imaging (WSI) systems to digitize and store them, which lacked FDA clearance for years.
But by the mid 2010’s, unbeknownst to me, things began to shift. Advances in whole-slide imaging technology (alongside FDA approval of them!), declining storage costs, and the rise of digital pathology—a term used to differentiate the analysis of scanned pathology slides from traditional microscopy—suddenly made the prospect of mass-collecting this data feasible. At first, it was primarily for communications sake; it’d be a nice convenience if a pathologist could easily ask a friend a continent over what they thought of a slide, or to digitally annotate a slide with their thoughts.
But more computationally trained people saw the potential value in doing something even more with this data. And so, startups in this space were founded around the same time as the rise of digital pathology, often spun out of collaborations with companies or hospitals who had their own gargantuan datasets of digitized pathology slides.
The two biggest names from back then are still around. There’s PathAI, which started in 2016 and had a network of 400~ pathologists to crowdsource pathology slide data. And then there’s Paige, which started in 2017 and had exclusive access to Memorial Sloan Kettering Cancer Center’s archive of 25 million digitized pathology slides.
Their thesis of these companies and companies like them was pretty simple: if deep learning could already outperform humans in tasks like object detection and classification, why couldn’t it do the same for pathology? Histopathology was, at its core, an image recognition problem—pathologists were trained to identify morphological patterns in tissue slides to diagnose diseases like cancer. AI models, given enough high-quality data, could potentially match or even exceed human accuracy in detecting tumors, grading disease severity, and even predicting patient outcomes.
Early research validated this idea. In 2017, a JAMA study demonstrated that deep learning models could identify breast cancer metastases in lymph nodes with accuracy comparable to trained pathologists. Google Health followed in 2020 with a model that could grade prostate cancer aggressiveness at sensitivity levels exceeding human experts. The stage was set for something very interesting to happen!
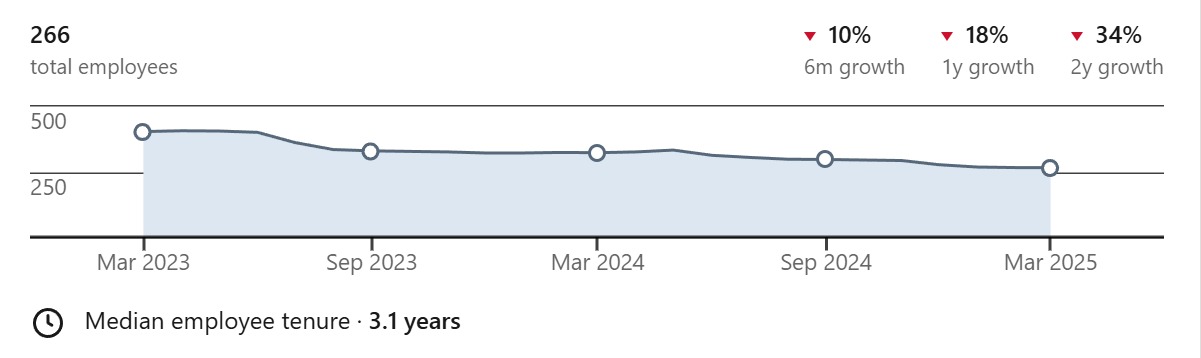
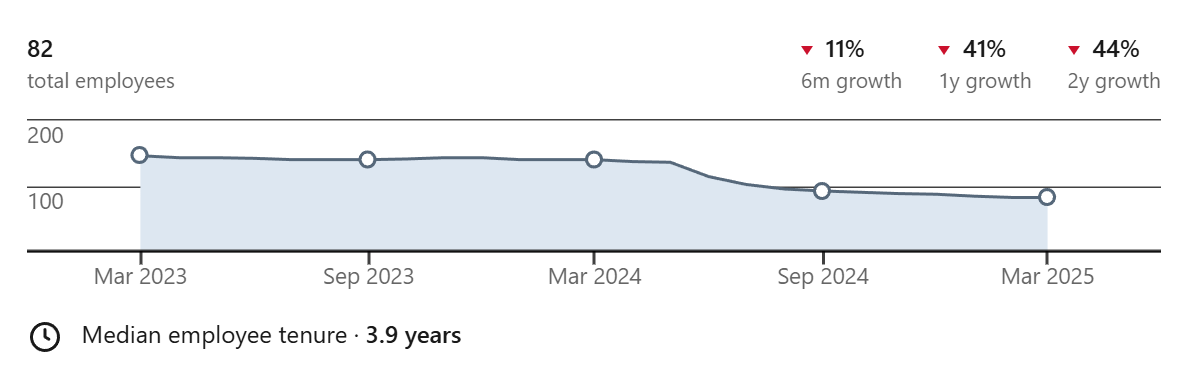
And yet…each of the companies I named earlier seem to have all the outward signs of doing badly. For one proxy, all have dramatically slashed their workforce.
Here is PathAI, dropping 34% in two years:
Here is Paige, dropping an even worse 44%:
Past that, their recent papers aren’t anything particularly impressive and I rarely hear about them at all anymore. Of course, workforce count, paper prestige, and being publicly visible aren’t necessarily tied to success at all. Downsizing, not caring about papers, and being private about internal affairs are all characteristics found in successful companies, but it does lead one to wonder.
This should lead us to an obvious question: what happened to pathology AI companies?
Did they succeed beyond their wildest dreams, quietly thriving in the background? Did they outright fail for reasons that are under publicized? Or, much like other biotech-ML companies, are they still stuck in an awkward middle ground, where progress is being made, but far slower than expected? And whatever the answer is, why did that happen, and not something else?
This essay will attempt to answer that question.
The problems
The death of traditional pathology was greatly exaggerated
This is, fairly, somewhat of a bait-and-switch on my end. Earlier, I mentioned that by the mid-2010’s, the digitization of pathology slides had really begun. And it had! By 2017, the first whole-slide-imaging system had been approved by the FDA, and by 2019, the second.
But approval doesn't equal adoption. According to one interview conducted in 2021, 56% of surveyed pathologists reported not having digital pathology infrastructure in their NHS hospitals. On a more anecdotal level, this particular Reddit thread from just a few months back was illuminating. Here are some quotes:
“I’m at a large state hospital and they use slides on a microscope”
“While digital sign out is being more widely adopted, the vast majority of practices are still looking at glass slides with a microscope.”
“In the US, 99% of pathologists are using traditional microscopes for diagnostic work.”
Why has been such little adoption of a seemingly useful tool?
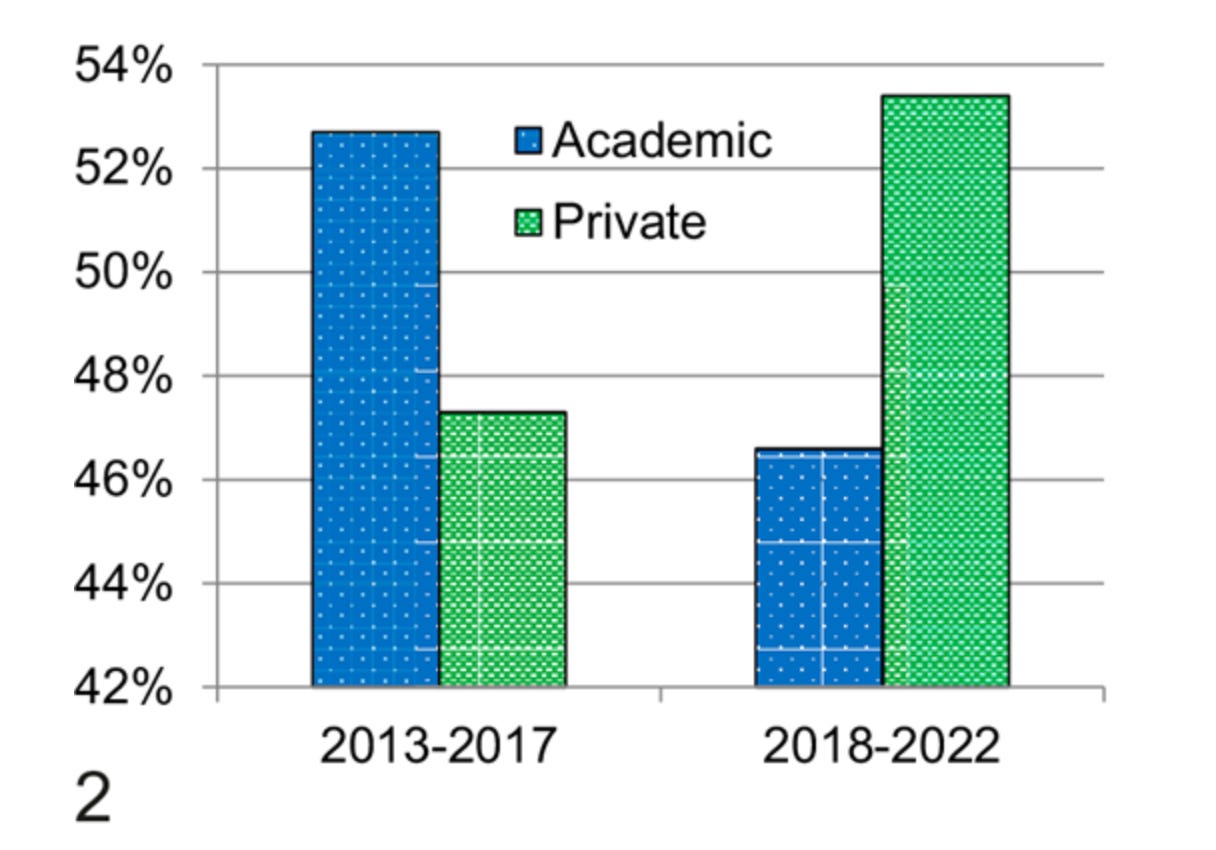
First, the economics are challenging. I mentioned that WSI scanners are expensive, but didn’t give exact numbers; a single whole-slide scanner can cost between $150,000 to $250,000 at the top end, with additional expenses for storage infrastructure, image management software, and workflow integration. For large academic medical centers processing thousands of slides daily, the initial investment can easily run into millions. This explains why pathologists employed by private companies spend far more of their time looking at digital slides than those working in the NHS (75% vs 25%). And, unfortunately for startups hoping to sell to people with digital slide workflows, there is a roughly equal number of pathologists in both groups.
Second, even for the groups that are able to afford a WSI scanner, pathologists still often don’t rely on them. This isn’t for luddite reasons, the software to navigate digital slides just…isn’t very good. At least if the bar for ‘good’ is ‘as easy as doing it by hand’. Remember, at least for now, humans must still be part of the pathology scanning process, so their opinions matter!
What are examples of software issues? Simple, common tasks on a microscope—quickly switching magnification, scanning across a slide, or comparing multiple slides side-by-side—became multi-click operations. Moving a glass slide under a microscope has zero latency, whereas digital systems introduce perceptible lag when navigating high-resolution images. Color artifacts can be introduced by digital displays. Logging in and out. And so on. For a pathologist reviewing dozens of slides daily, these small delays mean that they will be quick to abandon an otherwise theoretically useful system.
But let’s say you’re able to find pathologists willing to do digitization and actively rely on those digital slides. How do you make a business model with that? Well…
The right business model is…unclear
If you started a pathology AI company, you had roughly two choices on how to sell things to people.
The first option is to hire your own team of pathologists, armed with your in-house set of AI tooling, and tell hospitals and research groups to send their slides to you (no digitization needed), assuring them that you can get them results faster and cheaper than anyone else. In other words, services.
The second option is to simply pre-package the AI tooling itself and try to license it to whichever external group is already doing digital pathology analysis. In other words, products.
A lot of people chose the first category.
On paper, this made some sense. Vertical integration! Full control! You get access to your own stream of pathology data, you avoid slow enterprise sales cycles, and you can spin up a business that looks more like a tech-enabled lab than a pure software vendor. Given all we’ve discussed about how pathologists are loath to work with software tools, this wasn’t an insane path to go down. If the legacy businesses do not wish to adapt, simply compete with them on where they stand. If you believe in your ability to make efficient a difficult process, why wouldn’t you win? After all, it’s worked before in a related industry. It should work here!
Well, it’s not that cut and dry. Though there are some auxiliary benefits to trying to spin up your own AI-enabled pathology lab, there is a one small hurdle: in going down this path, you will, at some point, start competing with Labcorp and Quest. And it is really, really hard to win a battle with Labcorp or Quest.
To give some sense of the scale at which these two companies operate: Labcorp processes over 500,000 specimens per day. Quest clocks in around 400,000. They have nationwide logistics, massive economies of scale, and negotiated contracts with every major insurer. Their labs run like clockwork. Their margins may be thin, but they make it up in volume, efficiency, and market entrenchment. They have been optimizing clinical workflows for decades. They have fleets of drivers, entire warehouse-style labs optimized to process slides and bloodwork at scale, and enough regulatory experience to make new test approvals far less of an issue than it’d be for any upstart. They are not the slow-moving incumbent that one may mistake them as. They are a logistical marvel akin to Amazon that just happens to do pathology, alongside hundreds of other diagnostic tests.
And at least one pathology AI startup tried to compete with them: PathAI, who acquired Poplar, a clinical diagnostics pathology lab, in 2021. It feels deeply likely to me that PathAI believed that their AI capabilities would allow them to run a pathology lab more efficiently than the incumbents—that their models could streamline workflows, reduce error rates, maybe even lower costs per diagnosis. And maybe, in narrow ways, they were right. But at least from the outside looking in, the gains weren’t enough. Quest, in turn, bought Poplar from PathAI in 2024. Perhaps there was a multiple earned on this deal, but if clinical diagnostics wasn’t such a pain to deal with, why sell it off? This said, I’ve attached an alternative take on the situation by someone I talked to in the footnotes.1
Yet, a skeptical reader may have one response: for however bad a services play is, a products play feels worse.
Even in the best-case scenario where your product works perfectly and delivers some level of value, you are still at the mercy of institutions that adopt technology at glacial speed, have labyrinthine procurement processes, and often have no clear incentives to change anything at all. You're selling into healthcare with a contract, not just a one-and-done thing as with the services play. The end result would likely be that you might sell into a pilot. A small departmental deployment. But wide-scale enterprise adoption? That almost never arrives. Why would it happen? How much money is a pathology tool actually saving a hospital? Given that a fraction of the universe of pathologists have access to slide digitization, and that AI-based tools cannot by themselves sign off on cases given current regulatory guidelines…what’s the value pitch? You make things slightly faster? How much could that be worth? Empirically, the answer is not that much according to studies in the UK NHS system.
Well…unless your customer really does care about radical change, and where even minor workflow improvements are worth it. Unfortunately, that isn’t in most clinical systems. But it is in the private sector.
One pathology AI founder told me that it wasn’t hospitals or diagnostic labs that showed the most promise. It was R&D groups within Big Pharma. Those scientists and executives wanted new tooling. They were often sitting on massive internal datasets, had real budget allocated to experimental tech, and — critically — had a clear ROI if your model helped shave months off a study or more precisely target the right patient cohort. Most importantly, pharma didn’t care as much about the regulatory headaches, as they weren’t using your model to diagnose patients. In many ways, this was the path that Paige went down with their Omniscreen platform — helping pharma groups do better patient stratification, hopefully leading to better results in clinical trials.
But the markets there are small and growth is slow — there are only so many such R&D groups in the world that would want a product like this. So how could the increased margins be worth the squeeze? Past that, if ones pathology tool was so fundamentally useful, wouldn’t your customers just try to develop the tool internally? At least if you have a services play, you’d appeal to R&D groups that lack pathology digitization. But if they do have access to that, then isn’t it only a matter of time before they use their internal data to replicate your model?
So, between services and products, neither seems to be a particularly good category. Yet, empirically, some pathology startups have been able to make each individual category work. Consider Proscia, who just raised a $50M round and is a pure products play, and Artera, who is actively growing and is a pure services play. What’s going on there? We’ll discuss that at the end.
The value of the AI is somewhat questionable
Let’s assume you’ve solved digitization. You’ve figured out a workflow. Through an ungodly amount of effort, you’ve got a buyer or a partner or a lab. The slides are flowing in. You have clean, well-labeled data, and you can run your models without friction. And let’s even assume your model is well-calibrated and accurate, because, as of 2025, there are indeed off-the-shelf models that can rival expert pathologists in certain narrow tasks. Surely now is when the AI finally starts to deliver?
I think it’s worth considering that the average job of a pathologist is a far bit more complicated than simply ‘look at the slide and think’. A slide read is rarely an isolated act. It’s informed by clinical context, by knowledge of prior patient history, by awareness of what’s even being looked for in the first place.
Consider the following situation: a pathologist is reviewing a lung biopsy. The H&E stain suggests a possible adenocarcinoma, but there are ambiguous features—maybe some squamous differentiation, maybe not. The patient has a history of multiple primary tumors, and the oncologist has specifically asked whether this lesion is new or a metastasis. The pathologist pulls prior slides, requests IHC staining, and spends time correlating the imaging report. Only after that broader synthesis can a diagnosis be confidently rendered.
What exactly could the the AI contribute here?
Identifying areas of abnormality is helpful, yes, but it’s only a fraction of the interpretive task. The AI doesn’t have a way to reason within the workflow. It doesn’t correlate. It doesn’t ask follow-up questions. Which makes it easy to overstate how much of the workflow a model can actually own. Now, to be clear, could it? Could a model someday integrate clinical data, query prior records, suggest stains, and reason across multi-modal evidence? Absolutely! If given all of that context, I have relatively little doubt that such a model would be extraordinarily useful.
But, as far as I can tell, none of the models can do that, they poke at a very small part of the overall workflow: simply identifying areas of concern. A skeptical reader may shrug and say ‘sure, but isn’t replacing that small part still helpful?’. Maybe! And I’m sure there are some very specific niches in which replacing that slice of a pathologists workflow can indeed be useful. But given my earlier points about how much of a slowdown digital pathology tools often can be, it still feels like a hard sell.
One interesting note: unrelatedly to this article, I mentioned to a biotech AI founder — one who has worked with pathology slides before — that I was working on this piece, and what their take on pathology AI companies were. They barked with laughter and said ‘Have you seen how fast a pathologist works? A tool would need to outright replace them for it be a good value proposition to their employer.’. Maybe a useful datapoint to have as to difficulty of this problem.
Where does the industry stand today?
For all that I’ve mentioned about PathAI and Paige, the pathology AI industry as a whole doesn’t seem to be outright dead. At least, venture capitalists are still active in the area. A recent LinkedIn post highlighted a series of 10M+ funding rounds of a set of recently started pathology AI companies. And even if you look at Paige and PathAI, they haven’t outright disappeared. Struggling, but still alive!
So, what happened to pathology AI companies? Given the relative lack of literature on this area, I am forced to rely mostly on anecdotes from people I’ve spoken with, but I’ll call out two things.
One, the hopes and dreams of the prior generation of pathology companies can still be found in the upstarts — there hasn’t been some sort of reckoning that dampened anyones ambitions. But this may not be naiveté; whole slide imaging is accelerating year after year, and there will be some tipping point where the possible market for a computational pathology company really is quite sizable. Time will tell if we have yet reached that tipping point, but at least some people are optimistic.
And two, the business model problem and the value-of-AI problem has turned out to be somewhat related to each other in a pretty interesting way. As we’ve discussed, both services and products seem to be bad businesses, but yet some of the newer startups still seem to make it work. How? The answer may lie in the specialization of the AI they are developing.
For an adequate services play, you cannot merely have a ‘pathology foundation model’ that can do breast cancer screening or whatever. Instead, you must be able to offer a useful clinical service that no human pathologist can accurately do. Because otherwise, one of the bigger players will just eat your lunch through pure logistical power. Artera’s pathology tools for prostate cancer biopsies fits this. The results from this test include not only the typical clinical markets (e.g. Gleason score), but also how likely an intervention — specifically ST-ADT, or Short-Term Androgen Deprivation Therapy — would help the patient, judged entirely via AI applied to the pathology slide. A human pathologist cannot offer such an insight! And, importantly, the utility of such a metric was proved out in retrospective analyses of multiple Phase 3 trials.
For an adequate products play, your software must not only handle the application of AI to a digitized slide, but also handle…basically every single other part of a pathologist’s workflow. Proscia’s platform, Concentriq, is akin to this. It is closer to a full-stack digital pathology operating system rather than a ‘throw in digital slide and AI will be ran over it’ API. The product integrates slide management, collaboration tools, workflow automation, and ML tools into a single interface. For institutions already committed to digitization, this holistic approach can justify the cost, even if the AI itself isn’t actually all that helpful.
Of course, the problems for both sides will persist. For services, being limited by lab space can be annoying and your margins will be thin. For products, proving ROI can be a challenge, and growth is slow given that you have relatively few customers. And one issue will be faced by both groups: your competitors (services) or customers (products) are extremely eager to find some way to copy/replace you.
So, answering the question of what happened to pathology AI companies, a more succinct answer seems to be: they are still being proved out. There hasn’t been a clear winner as of yet, but the losers didn’t arise from some fundamental flaw with the underlying technology. This is a boring answer, given that the same thing could be said about nearly every AI biotech, but it does seem to be the correct one. ML applied to pathology is undoubtedly the future — as is ML being applied to everything — but it feels like we’re still in early innings for the whole process, even a decade after the first companies in the space popped up. I do get the sense of a ‘we’ve learned what doesn’t work’ from people in the field, which I consider to be a hopeful sign.
Of course, this essay is very much an oversimplification. Digital pathology is an insanely large space, and while I think my coverage of the area does cover a sizable fraction of it, I am undoubtedly missing some. If you’re working in this space and think I’ve missed something, I’d love to hear from you!
Well, one last thing that I couldn’t quite fit in anywhere else. Mostly because it’s somewhat unrelated to the point of this essay, but it’s an interesting tidbit.
A phenomenon I’ve long been a fan of in the biotech space is the existence of startups that exist to solve some incredibly hyper-specific bit of a workflow, one that you’d never even think about. Plasmidsaurus is my canonical example of this, plasmid Q&A wasn’t something I ever assumed was a big deal. But I didn’t consider that there was such a company in the digital pathology world — everyone here seemingly wants to solve everything, or at least a very broad category of problems.
But I did stumble across one startup that does fit the bill: Pictor Labs. The thesis of this company is virtual staining — using AI to computationally generate histological stains (like H&E, trichrome, or IHC markers) from label-free or minimally stained tissue images. Pictor’s premise is simple: physical staining is slow, expensive, and destroys tissue. Traditional staining requires slicing tissue into multiple sections, applying chemical reagents (some toxic or proprietary), and waiting hours or days for results. For rare biopsies, this wastes precious material. For labs, it’s a logistical headache. Pictor’s AI models instead take a single unstained slide and predict what other stains would look like if applied.
Simple! Saves time! And money! This is ostensibly a software product, but it has the ‘we’re doing something nobody else can’ services vibe to it. Very cool and something I feel like will make their founders quite rich via an eventual buyout. But it’s not really worth mentioning in the grander scheme of this essay, as Pictor just launched in 2019 and don’t seem to have any (public) partnerships, so perhaps there is some flaw here I’m not seeing. But I found the pitch endearing enough to mention here. Hoping for more startups in this vein!
“In my opinion, PathAI acquired Poplar, not for clinical workflows but for the data play -- build the algorithms for diagnostics + research (drug development). The clinical workflows was just the short term play that would allow them to also find stickiness and test things in iterative cycles.”





I know a very well-positioned founder working on launching something quite exciting in this space, if anyone reading this is curious for an intro
The Paige.AI story is also interesting due to some high profile controversies with their relationship to MSKCC and some recent cofounder exits.